A staff member displays a sample of the COVID-19 inactivated vaccine at a vaccine production plant of China National Pharmaceutical Group (Sinopharm) in Beijing, capital of China, April 10, 2020. (Xinhua/Zhang Yuwei)
KATHMANDU, June 5 (Xinhua) -- Nepal's drug regulator has approved the COVID-19 vaccine developed by Chinese pharmaceutical company Sinovac Biotech for emergency use in the country, which is suffering from a deadly second wave of the pandemic.
It is the second Chinese vaccine to have got approval from the Department of Drug Administration for emergency use in Nepal.
Nepal has already granted approval to the COVID-19 vaccine developed by China's Beijing Institute of Biological Products Co. Ltd, under Sinopharm, for emergency use in Nepal.
"It is to notify to all concerned that the conditional permission has been granted for emergency use authorization of COVDI-19 Vaccine (Vero Cell) Inactivated (CoronaVac) manufactured by Sinovac Life Sciences Co. Ltd in Nepal," said a statement issued by the department on Friday.
According to the department, the vaccine was approved for immunization of individuals aged 18 or above.
Bharat Bhattarai, director general of the department, told Xinhua on Saturday that the authorization was granted to Sinovac after the regulator completed the study of the data provided by the Chinese company.
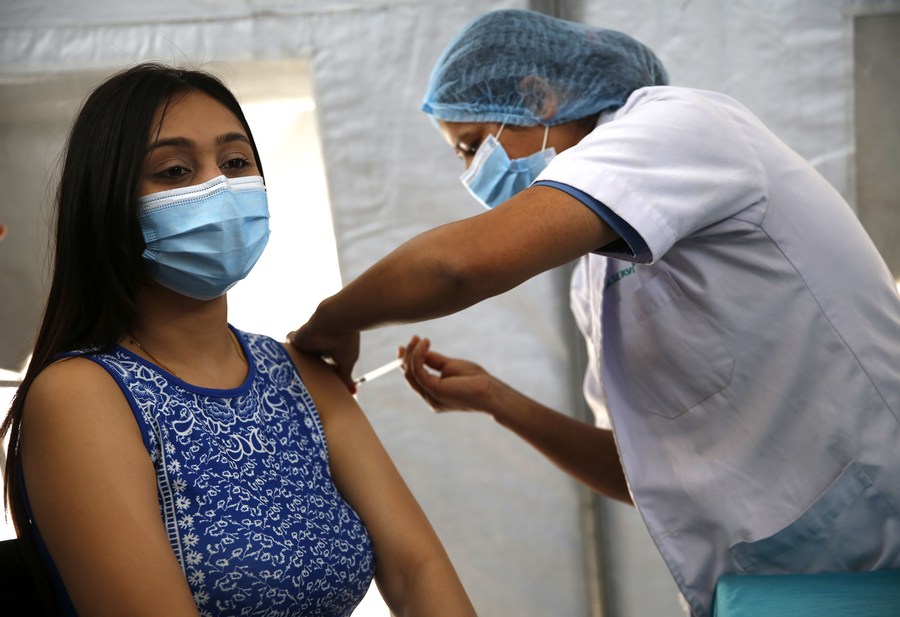
A girl receives the first dose of COVID-19 vaccine at a hospital in Kathmandu, Nepal, on April 7, 2021. (Xinhua/Sulav Shrestha)
"It is also the last vaccine to get regulatory approval from the department. We have not received an application from any additional company for approval at the moment," said Bhattarai.
On June 1, the World Health Organization validated the Sinovac vaccine for emergency use for people 18 years and older.
In face of the raging coronavirus, Nepal has only got 2 percent of its some 30 million population administered twice for now as there is a short supply of vaccines, according to the Health Ministry.
As of Friday, the South Asian nation had recorded 581,560 confirmed COVID-19 infections and a death toll of 7,731. ■




 A single purchase
A single purchase